Our Research
The Macé lab addresses how brain-wide networks interact to produce a particular behavior. We use functional ultrasound imaging, a technique we pioneered, to follow neuronal activity in the whole mouse brain. We combine this technique with targeted genetic circuit manipulations and electrophysiological measurements. One goal is to better understand how cognitive processes, such as behavioral switching, arise in the brain across multiple scales and uncover dysfunctions of these networks in psychiatric disorders. Our lab also studies visual processing and develops innovative optogenetic strategies against blindness to restore visual perception and visuomotor function.
Latest Publications

A transition-prone brain state precedes spontaneous behavioral switching
bioRxiv, 2026
Here, we studied the initiation of uninstructed behaviors of head-fixed mice in two contexts: a virtual burrow and a running wheel. We employed functional ultrasound imaging (fUS) to record whole-brain activity and to identify whether the initiation of spontaneous behavior could be predicted from hemodynamic signals. We first identified distinct hemodynamic patterns associated with each behavior and subsequently performed time-resolved decoding to predict behavioral transitions from fUS data. We found that whole-brain hemodynamic signals could decode spontaneous egress and running around 10 seconds before their onset, a timescale that cannot be accounted for by preceding behavioral changes alone. Furthermore, we found a network of regions, including the medial septum (MS), that decreased their signal several seconds before the onset of egress and running. Mimicking this decrease by inhibiting neurons in the MS via optogenetics increased the probability of egress, running, and grooming. Through this unbiased approach, our work sheds light on a whole-brain transition-prone state that precedes uninstructed behavior transitions.

Arousal elicits a brain-wide hemodynamic wave independent of locus coeruleus noradrenergic tone
bioRxiv, 2026
Arousal fluctuations during wakefulness have a major impact on physiology and behavior, including perception and task performance. Arousal is also known to be a strong modulator of neural activity, but the brain-wide spatiotemporal structure of this modulation is not fully characterized. We used functional ultrasound imaging to record brain-wide hemodynamics – a proxy for neural activity – in head-fixed mice during spontaneous and sensory-evoked arousal fluctuations, tracked via pupil diameter. Both conditions recruited a common brain-wide hemodynamic wave that followed a subcortex-to-cortex gradient. We then tested whether noradrenaline, widely associated with arousal, was necessary or sufficient to drive this wave. Sustained bidirectional optogenetic manipulations of locus coeruleus activity affected brain wide vascular signal amplitude but, surprisingly, left arousal-linked dynamics largely intact. Together, these results identify a common spatiotemporal motif of arousal that appears independent of noradrenergic tone.
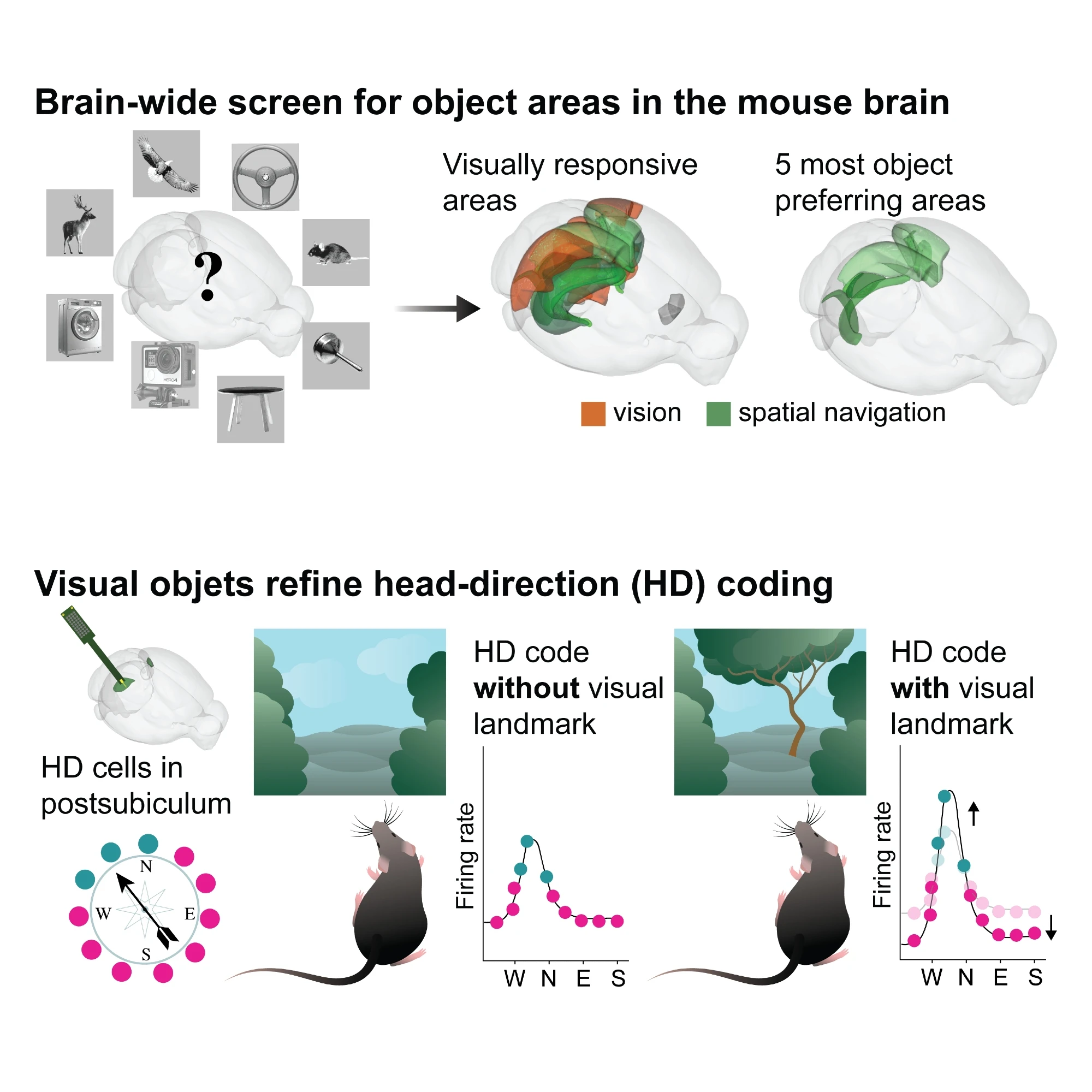
Visual objects refine head direction coding
Science, 2025
Using functional ultrasound (fUS) imaging in mice, we conducted a brain-wide screen and identified brain areas that were preferentially activated by images of objects compared to scrambled versions of the same stimuli. While visual cortical areas did not show a significant preference, regions associated with spatial navigation were preferentially activated by visual objects. Electrophysiological recordings in the postsubiculum, the primary cortical area of the head direction (HD) system, further confirmed a preference for visual objects, which was present in both HD cells and fast-spiking interneurons. Finally, we found that visual objects dynamically modulate HD cells, selectively increasing firing rates for HD cells aligned with a visual landmark’s direction, while suppressing activity in HD cells coding for other directions. These results reveal that visual objects refine population-level coding of head direction.